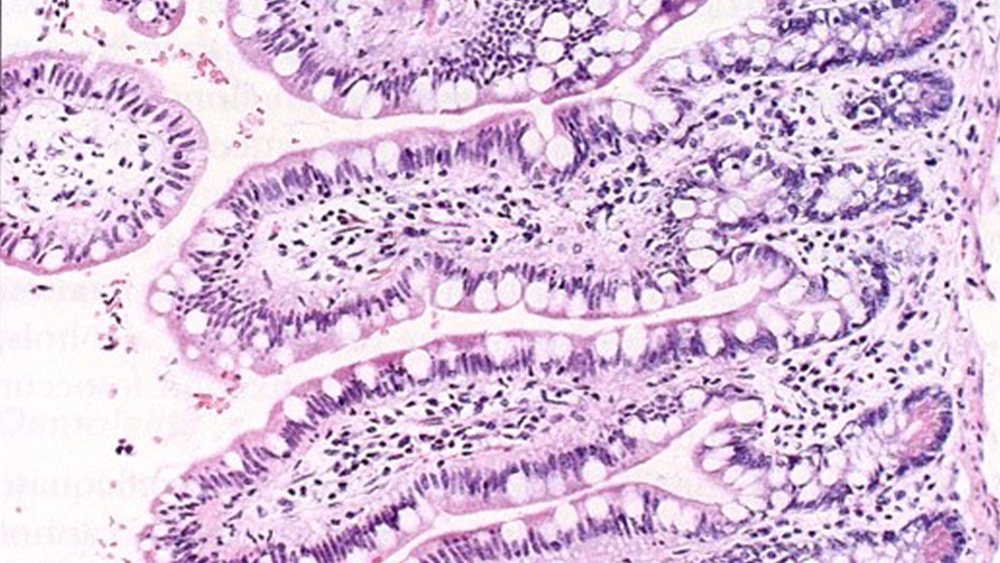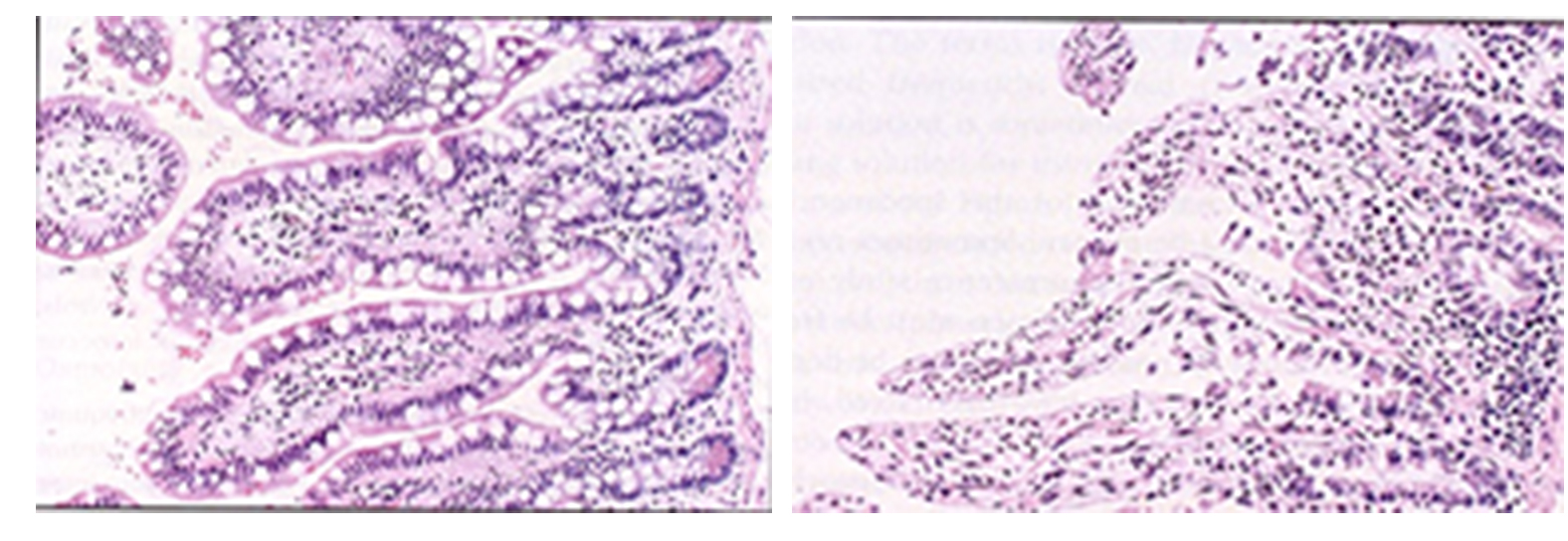
One of the primary fixation/processing artifacts noted in laboratories by the NSH/CAP HistoQIP quality assurance assessment program, is over/under fixation. The images below demonstrate the effects of delayed fixation where tissues where not placed immediately in formalin fixative, allowing the process of autolysis to begin. The image on the left shows a small intestine tissue specimen placed immediately in fixative. Note the presence of the columnar epithelial layer on the intestinal villi showing very distinct goblet cells. This section is well fixed and preserved for anatomic study. The image on the right demonstrates delayed fixation. Note the complete absence of the columnar epithelial layer. This has been stripped away by the natural enzymatic activity breaking down cells once the blood supply has ceased. This tissue has been destroyed and rendered useless to the pathologist in diagnosing disease.
The key to maintaining optimized fixation is process and consistency. Every lab is challenged to develop and validate effective fixation procedures for the tissues they work with and the fixatives they use. Developing a procedure begins with taking the aforementioned knowledge of penetration rate and time and applying that to a universal procedure that can accommodate ≥ 90% of the tissues you process. In a hospital setting this would include all the main tissues and organs (kidney, liver, muscle, upper and lower GI tract, etc.). The procedure should be able to be applied to standard use for these tissues, if standard grossing procedures are maintained at 3-5mm thickness of specimens grossed. The exception within the 10% margin would be fatty specimens (breast, colon, adipose tissue, blood clot, etc.). These may require longer fixation procedures with additional solution changes depending on the degree of fat. The other exception within the 10% would be biopsy specimens, which would require shorter fixation times. Bone specimens are another exception which would require a specialized procedure of longer time due to its dense nature.
As previously mentioned, specimens should be grossed at a mandatory thickness of 3-5mm and should be placed in a volume of fixative at a minimum of 20:1 fixative to tissue volume. It is imperative to have an excess of fixative fluid volume to tissue volume because with some fixatives, particularly 10% neutral buffered formalin, the key ingredient of the fixative becomes depleted in the process of molecular changes in tissues. Communications of this requirement between the Histology supervisor/manager and any/all departments or facilities submitting specimen is necessary. This is to avoid the frequent problem in some hospitals of the surgery department transporting specimens that are not even covered in fixative, let alone the volume of 20:1 required for fixation; and also with some offsite physician office laboratories. The total time involved in the fixation process should be in a range of 4-24 hours depending on the size, nature, and density of the specimen. Small biopsy specimens including needle core biopsies, fine needle aspirations, bone marrow core biopsies, etc., can be fixed in a shorter time of 2-4 hours depending on the specific type of tissue.
Understanding that the fixative reagent can quickly become diluted with repeated use and thus not reliable or consistent from one tissue batch run to another, it is critical to change or at least rotate fixative solutions on a routine basis based on the number of tissues run through the solution or the number of days the solution is used. Each lab must determine what solution changes/rotations are necessary to maintain the effectiveness of the fixation process. If a lab is running high volumes of specimens or a large amount of fatty specimens, it is advisable to change or rotate solutions daily with at least 2 changes of fixation reagent. As a standard quality control monitor, Q.C. charts should be developed to record and document daily routine changes of fixatives. These charts should have a sign-off for the lab personnel maintaining the instrument, and the charts should be monitored daily to insure verification of maintenance and potency of fixative solution. Technologists should work closely with their pathologist to develop a protocol that effectively fixes general tissues in the lab and is reproducible from trial upon trial.
Once a protocol has been developed, it must be validated and documented ‘before’ being adopted into the standard operating procedures (SOP) and daily use. The validation process entails taking an adequate representation of tissue samples done in the lab and taking them through repeated trials of the fixation protocol followed by the standard tissue processing embedding/microtomy and H&E staining procedures. Each trial is reviewed by the lab supervisor or designated personnel, then by the pathologist. The pathologist will then sign off on this, and only then is it in compliance to be used as a standard procedure on patient specimen.
With having developed and validated an effective protocol, the success of this is now totally dependent upon consistency. Respectfully speaking, there is no excuse in patient care for not monitoring and maintaining these processes on a daily basis. Not having enough personnel; not having time on certain heavy days; allowing lab personnel to skip this maintenance for any reason; none of these are acceptable reasons to put patient specimens at risk and compromise the ability for diagnosis. Many steps in the tissue processing process (dehydration, clearing, infiltration) can be reversed if not performed optimally, but not fixation. Fixation is permanent and incomplete or poor fixation ‘will’ affect the efficacy of the proceeding steps in processing. Further, it ‘will’ affect subsequent routine H&E or advanced (special staining, IHC, etc.) staining. The importance and necessity of this daily maintenance ‘must’ be emphasized to the staff personnel and daily Q. A. monitors (charts) ‘must’ be monitored. Any deviation from the daily maintenance should be addressed and corrected immediately. It is a simple process that must be maintained through a commitment of personal integrity and responsibility to the patient.
References
Brown, RW; ‘Histologic Preparations: Common Problems and Their Solutions, College of American Pathologists, Northfield, IL., 2009.
Brown, S., “The Cost of Reprocessing”, Professional Development Series, Lab Management Consultants 2018.
Carson, FL, Histotechnology – A Self-Instruction Text, Chicago, IL., ASCP Press, 1997.
Hewlett, B., “NBF Penetration Rate”, Histonet Server, 2002.
‘Tissue fixation/Volume of fixative’; Internet search, www.bing.com.
www.NSH.org.