A quote from a well-respected teaching manual in Histology states, “Fixation, the stabilization of proteins, is the singularly most important step in producing good histologic slides”. Further, “The finished microscopic tissue slide, on which a diagnosis may be made, can only be of value to the pathologist if the tissue is properly fixed”. Given that it is a natural process for living cells and tissue to break themselves down upon cell death by a process called autolysis, fixation can be thought of as the foundation of anatomic pathology preservation and study. Many times tissues that are not optimally, or only partially fixed, will still render morphologic structures enough to diagnose disease, but the ability to view, assess pathology, and determine how far a disease has spread is drastically compromised if the tissue is not properly, even optimally fixed. Understanding the purpose and function of a good fixative is the beginning of optimizing tissue preparation.
Enzymes are certain proteins within the body that begin to break the cell down upon cell death. Fixation suppresses or halts this process and literally fixes the cell in its current anatomic state. This allows subsequent tissue preparation procedures to be done without the ongoing degradation of the cell. Fixation renders the cellular components (such as nuclei) insoluble and resistant to the effects of tissue processing steps. The tissue is thus preserved in a state closest to its original state and presents a picture to the pathologist of disease and its manifestation into normal tissue.
A good fixative will somewhat harden the tissue by the removal of bound water and maintain the relationship between cells and extra-cellular substances. The two diagrams to the right demonstrate bound water on tissue molecules before and after fixative is applied.
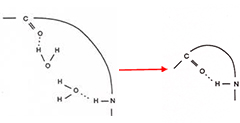
This is part of the hardening effect of fixation. For fixatives to be effective they must be used relatively fresh to make sure that their enzymatic suppression works optimally. The more times a fixative is used, the weaker the fixative becomes, and its ability to stop autolysis decreases. The volume of fixative to specimen size should be a minimum of 2:1, but for longer fixative methods it should be a higher volume such as 3:1 or 4:1.
Penetration rate and binding time are two additional factors that determine the effectiveness of a fixative. Tissue specimens should be optimally grossed at 3-5 mm to allow penetration of the fixative solution. Additionally, penetration rate of formalin fixative begins at a progressive rate of 3.6 mm per hour, but slows down progressively with time.

In 4 hours it will have progressed to only 7.6 mm. and will continue to slow down. Specimens that are grossed thicker than previously mentioned will slow the penetration rate even further. Some form of motion of the specimen in the fixative solution, such as agitation or swirling, can enhance the penetration rate and ensure complete penetration. We see this agitation methodology built into most of the tissue processor instruments in the market today. Even when the specimens are optimally grossed at 3-5 mm., there is a ‘binding time’ that is necessary for the chemical reaction of fixation to complete its process. Formalin is in the class of fixatives called ‘additive’ fixatives, meaning that it chemically links to the tissue molecule altering the structure. In the process an additive compound is formed linking it to other tissue molecules. Thus the molecular structure is changed in the fixation process to stabilize the enzyme proteins. This process takes optimal time; thus time in fixative is equally if not more important than thickness of tissue. The image on the right shows one of the effects of tissue that was either too thick and did not allow complete penetration, or was not given enough time in the fixative solution to allow the histochemical binding process to happen. You can see good fixation on the tissue perimeter, but not in the center.
In the daily rush of clinical histopathology (hospital), it is a surprisingly common occurrence to have optimal fixation steps compromised by high patient surgical case volumes, inadequate number of P.A.s or pathologists to handle the grossing for this volume, late in the day surgeries, and even unreliable courier service schedules where tissues don’t arrive on time. Many hospital administrators, even doctors, are sometimes unable or unwilling to devote the necessary resources and expense to ensure these variables are addressed and the appropriate resources are available. In some cases hospitals have traveling Pathologists or P.A.s that cover multiple sites, meaning that on busy days the latter site(s) could get pushed back late in the day and not allow enough fixation time before tissue processing. In these cases (hospitals) we often see a high volume of reprocessing done, meaning the tissues must be redone causing delay in patient diagnosis, added cost to the lab, and added difficulty to the pathologist because of tissue degradation.
Optimizing your fixation process can be achieved by focusing on five major principles: 1. Size of the specimen. 2. Fluid volume ratio, 3. Temperature (room temperature or slight heat), 4. Agitation, and 5. Time. Tissues that are not properly fixed during the tissue processing process, never renders the same degree of diagnostic visibility to the pathologist upon reprocessing. In essence, failure to address and monitor something as critical as initial fixation ‘will’ compromise patient diagnosis. As health care professionals let us always strive to pay attention to the small things in our process. Taking care of the small things on a daily basis will avoid some of the big things that will be needed to take care of the problem created.
Lab Storage Systems has a complete product line of formalin fixative containers, empty as well as pre-filled. For more information contact our Customer Relations staff today.
References:
- B. Hewlett, ‘NBF Penetration Rate’, Histonet Server, 2002.
- Carson, FL, Histotechnology – A Self-Instruction Text, Chicago, IL, ASCP Press, 1997.
- Bancroft, JD, Gamble, M, Theory and Practice of Histological Techniques, London, UK, Churchill Livingston Press, 2002.
- Sheehan, DC, Hrapchak, BB, Theory and Practice of Histotechnology, Columbus, OH, Battelle Press, 1980.
- Lab Management Consultants – Professional Development Series for Laboratory Professionals, 2005.