In the previous series titled, “Understanding H&E Staining”, it was stated that high volume H&E staining laboratories would experience some degree of moderate to significant loss of stain intensity during the course of their daily run. Therefore, these labs would probably need to have some form of dye/reagent rotation during their staining series. In this addendum to that series there are two quality control monitors that should be developed as part of your H&E Quality Assurance plan:
- Quality control sheets that show the daily maintenance of dye/reagent changes and/or rotations.
- Daily quality control screening of equal to or greater than 10% of H&E slides stained for the day.
The Q.A. monitor for daily stain maintenance (#1), should be designed for one or multiple persons servicing the staining unit throughout the course of a day. Given that the lab would be a high-volume user, it would be very likely that the staining runs continued beyond the shift for one person. A simple table developed as a Word document could provide columns with all the mandatory data necessary for maintenance documentation such as:
- Number of slides per rack (this is how you would calculate your total slide count and know when dye/reagent change and/or rotations would be warranted.)
- The time a rack was placed onto the stainer. In addition to documenting when a slide was started or a dye/reagent adjustment5 was made, this would also provide a workflow assessment of your H&E slide production.
- Reagent refresh columns would document what specific dye/reagent was adjusted and if it was changed or rotated, e.g., Hematoxylin Changed, 95% Alcohol Rotated, etc.
- Initials of the person providing the stainer maintenance of the adjustment made.
Regulatory guidelines state that laboratories must have a schedule to change solutions in the automatic stainer at intervals appropriate for the lab’s work volume. Further, as evidence of compliance, there must be a written procedure defining the exact frequency of solution changes. The Q.A. document example shown (see table at the end) is for the Prisma High-Volume Stainer and demonstrates how every aspect of stain maintenance can be documented. This unit as well as other high-volume stainers typically have some form of slide count calculator within the system, however, an additional hard copy document is necessary to give specific information such as the frequency of each dye/reagent change and/or rotation. Every lab will be somewhat different based on the specific protocol they have validated for their H&E stain. The document created should also show the document reviewer, usually the supervisor or manager that oversees and validates that this was the stain maintenance done for that day. The document is then retained in a file and kept for verification during any regulatory inspections.
Q.C. monitor #2, ‘Quality Control Slide Screening’, also satisfies regulatory requirements concerning a random quality control check of a certain percentage of slides produced daily. While some regulatory agents vary somewhat, it is a general requirement that 10% or more of slides be selected to represent the general quality of staining produced. In many laboratories, this is highly problematic due to staffing restrictions and technical competency. In most Histology laboratories they don’t have the luxury of having one individual assigned to slide Q.C., because in a high-volume environment this would require considerable time away from other time-sensitive duties such as microtomy. Many labs relegate this responsibility to the case assembly or slide send-out person, the supervisory, or some other individual not involved in embedding, microtomy, or other priority assignments.
Nevertheless, it’s a regulatory requirement and moreover an absolute necessity for a laboratory Quality Assurance plan. Not only does it monitor the quality of staining and allow for immediate intervention when there is an issue, but it also helps monitor the quality of microtomy and address technique or artifact issues that may hinder or compromise diagnoses.
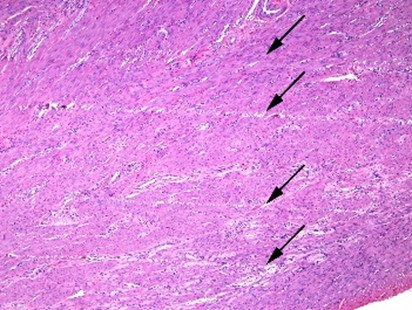
Common microtomy artifacts such as venetian blind effect, thick & thin sections, knife marks, etc., can be caught and the information conveyed to the microtomist so that immediate adjustments can be made. High-volume labs are frequently challenged by this because speed and production can sometimes compromise quality. This kind of Q.A. monitor helps maintain an acceptable union between efficient production and optimal quality. The image shown here is an example of discrete knife lines that demonstrate the microtomist should possibly change out their blade more frequently. While some of the knife marks are clearly visible, other artifacts known as ‘angel hair’ marks can only be detected under microscopic examination.
Adoption and routine practice of the two Quality Control monitors discussed in this article will help maintain the routine delivery of quality H&E slides to the pathologist for diagnoses. Daily practice will also ensure day-to-day consistency and give the laboratory continual updates on quality throughout the course of your daily staining runs.
- Brown, S., “Developing A Competency Assessment Program”, Professional Development Series, Lab Management Consultants, 2017.
- Brown, S., “Validating Your H&E Staining Systems”, Technical Series, Lab Management Consultants, 2019.
- College of American Pathologists (CAP), General Checklist – ANP.21360, 2020.
- Hansen, S., Images in H&E Staining, LBS. 2011.
