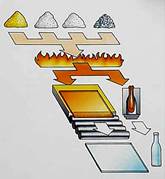
Since the late 18th century glass slides have been a major platform for microscopic and diagnostic study of plant and animal morphology. The translucency of the glass material made it a perfect mounting structure for samples where light could be refracted through and allow external and internal visualization of the specimen. The composition, formation, and manufacturing process of glass in general is not that complicated, however, it is a very lengthy and time-consuming process. The image to the right is an example of a flat glass manufacturing process where sand is heated at extremely high temperatures for extended hours. Sodium carbonate is added to reduce the sand’s melting point (2930º F) and save time and energy during the melt-down process. The molecular composition of the sand reconfigures to a product that would break down in water. Limestone is added to prevent this reconfiguration and render a product that in a molten state can be shaped and formed into any number of shapes then cooled. This is soda-lime-silica glass. Specifically with slides, the molten liquid is poured out onto thin, molten tin sheets, cooled, quality inspected, then cut into the desired parameters.
Medical applications such as Histopathology required a glass product that was not only clean and highly refractive, but also having a surface that would have an affinity for tissue and cellular specimens. Glass is by nature chemically inert and has relatively no natural attractive bond on its surface. Slides have little to no natural affinity to cellular or issue bonding, and therefore are highly susceptible to cells/tissues lifting off the surface if no physical or chemical treatment is applied. Heating or ‘baking’ mounted tissues on slides was a physical treatment that provided a weak initial attachment, but not enough to reliably guard against tissue loss. It became evident that some form of chemical bond needed to be applied to the surface to provide a bond strong enough to withstand the chemicals involved in cellular/tissue staining and rinsing procedures.
The evolution in the diversity of slide adhesives was driven by the specificity and sensitivity of the stain application used. When the major focus in early anatomic research and discovery was on basic morphology, glass slides were coated with some form of protein-based solution that would provide a nitrogen/hydrogen positive bond that would give the surface a stronger affinity for the nitrogen/hydrogen negative bonding in tissue.

This author’s first experience with this was as a new technologist being instructed to add Elmer’s glue either to the waterbath or directly applied to the slide and smeared with the finger to give a thin coating. This was done primarily with basic or routine staining procedures such as hematoxylin and eosin to visualize normal v/s abnormal cells/tissues. An inherent natural artifact that came out of this was that hematoxylin has an overall positive charge, which has an affinity for the negatively charged protein-based additives. Stained slides demonstrated a faint blue tint artifact on the glass, which with basic histopathology was just a mild inconvenience to the pathologist. But with special stains of greater diagnostic specificity such as silver stains, and particularly with the increase in immunohistochemistry studies, adhesive products and methods that had little to no background staining had to be employed.
Continued evolution of adhesives transitioned from superior adhesives to more permanently coated positively charged solutions such as poly-L-lysine, APES polymer coated, and others. Researchers discovered that a permanent positive charge could be applied to the slide via such types of solutions, and for the most part eliminate the problem of endogenous non-specific binding on the glass and significantly decrease the incidence of tissue lifting/loss. Commercial manufacturers began developing their own proprietary processes and solutions to create a hydrophobic surface that repelled water and helped decrease the water barrier between tissue and slide; transitioning even further to a hydrophilic surface that created more bonding sites for glass and tissue. This was all done to compliment the previously mentioned intense scrutiny applied to the original glass manufacture. Technologists now have a wide diversity of preferences for superior ‘charged’ slide adhesives and can now streamline their staining methods and procedures while enhancing their quality assurance on diagnostic tests.

Lab Storage Systems offers slides produced from the highest quality white glass for a multitude of histology, cytology, and hematology applications. Also, we have a complete line of non-coated, coated, positively charged slides, and specialty designed slides to address your specific laboratory. For more information or samples contact our Customer Relations Manager or Technical Marketing Specialist today.

References:
- Carmeuse, ‘Glass Manufacturing’, carmeusena.com, 2018
- Quill.com, Elmer’s Glue-All adhesive products.
- Woodford, C., ‘Glass’, Explainthatstuff.com, June 2018.

