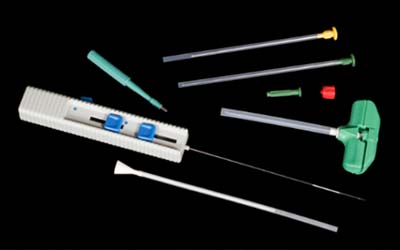
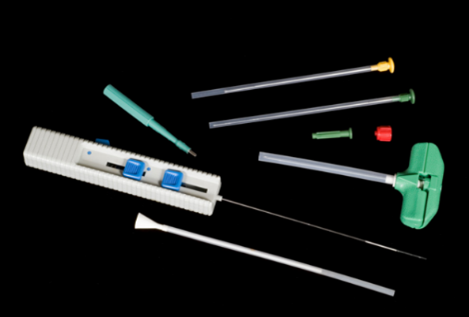
In the world of Histopathology, seldom do we still see the need for large, hunk-like specimens surgically removed for patient diagnoses. In the mid-2000s the profession began to see a transition from sizeable specimens that would consume a considerable majority of a tissue cassette, to smaller minute specimens such as needle core biopsies. Biopsy-like specimens proved to be more specific to the core of the tissue pathology and marginal areas, less invasive to the patient, and causing less trauma to the body and a quicker recovery. With these smaller specimens came the added responsibility and mandate for effective devices that would ensure the acquisition and retention of even ‘snippet’ like specimens, throughout the specimen handling and processing events. Surgical instruments such as those illustrated in this image were designed to perform tissue extraction.
These methods were further perfected by the onset of x-ray and other radiography types of assisted procedures. Once tissue extraction was achieved, specimen retention and maintenance of tissue integrity became the priority. This focus required thought and design modifications in not only tissue cassettes, but also materials within the cassette that would further ensure tissue was not lost anywhere in the process prior to being embedded in paraffin wax.
As seen here in Image 1, specimens are placed in the paper and the paper is folded over 1-2 times to hold the specimen. The paper holds the tissue well, but it does not do well when tissue debris needs to be filtered from a fluid medium (such as in fine needle aspirations). Also, the material of the lens paper is porous but does not promote fluid exchange as well as nylon biopsy bags or sponges.

Image #1 – Lens Paper
Image 2, to the right, shows a nylon mesh biopsy bag on top of a tissue cassette. This item is ideal for small minute specimens and even particulate matter debris-like tissue in a mucous or fluid medium; so fine and granular that it must be filtered into the bag. After processing, the bag would require gently tearing open the bag (so that no tissue debris would pop away) and scraping all contents into a base mold for embedding. The challenge to this (as well as lens paper) is that the bag must be continually held down on the hot plate of the embedding center while scraping to keep the paraffin from solidifying and the tissue adhering to the bag.

Sponges, also known as biopsy pads, (Image 3), are the most economical of the three specimen retention items shown. They secure tissues well, provide the most optimal mechanism for fluidic flow and reagent exchange in tissues, and offer a good contrasting background to tissues for better location during embedding. Two pads used to create a sandwich-like scenario with the tissue provides optimal security for retaining tissue. They are far less cumbersome to work with at the embedding center and easily lay flat on the hot plate keeping the paraffin melted around the specimen. One noted disadvantage is that they cannot be used with very fine particulate tissue (debris) because the tissue will filter down and become trapped in the sponge. Also, with some tissues, the sponge can leave an indention artifact on the tissue surface.

In this article, we have discussed the concept of specimen retention and the mandate for effective utility devices for tissue acquisition, security, and maintenance of tissue integrity. The three examples shown have been discussed with their respective strengths and weaknesses. But the decision of what is the best item or items depends on what is best for your laboratory and the tissues you most commonly work with. In many cases, laboratories use two or more items based on that specific tissue being processed (core specimen, filtered medium, etc.). One of the absolute prime directives in Histopathology, especially when dealing with patient specimens, is that tissues can never be lost in the process. As health care professionals, our responsibility to the patient is to employ whatever methods, procedures, or devices necessary to ensure uncompromised diagnoses and optimal patient care. In Part 2 of this series, we will discuss this mandate for retention as related to optimal tissue cassette design.
Reference:
- McCormick, J.B., “Snippetology-The Science of Small Specimen Management”, Science Heritage LTD., 2006.
- Brown, S., “Small Specimen Management – In A Large Volume World” , Lab Management Consultants Educational Development Series, 2008.
- www.labstore.com
- www.carolina.com
- www.globalscientific.co.uk