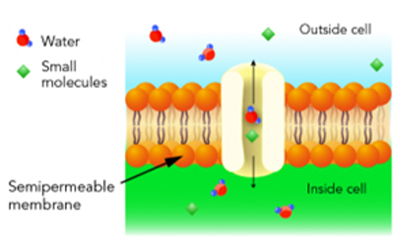
All tissue is made up of one or more cells. Cells are the fundamental and structural units of life that give various tissues their specificity (liver, kidney, muscle, etc.). The cell membrane, or protective border around the cell, encapsulates all the organelles (nucleus, mitochondrion, endoplasmic reticulum, etc.) in a cytoplasmic medium. These organelles are the things that function together to give the cell life. The cellular membrane is actually a ‘semi-permeable’ membrane allowing certain fluids to flow back and forth in/out of the cell.
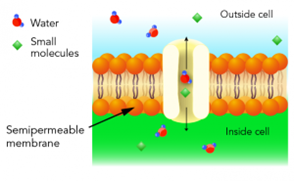
Referring again to the image and understanding the process of tissue processing where water has been removed from the cell through the alcohol dehydration, and the alcohol then cleared out with xylene; the cell is then interstitially coated with wax through infiltration. We have now created a wax barrier to the outer (blue) and inner (green) portions of the membrane. This wax barrier on the membrane is enhanced even further by tissue block embedding. The obvious reality is that, after microtomy, before any slide staining (routine or special stain) is possible, this wax barrier must be thoroughly removed. Further, after the wax has been removed (deparaffinization), the tissue cells must be hydrated with water to give the cellular medium and organelles the affinity to absorb staining dyes.
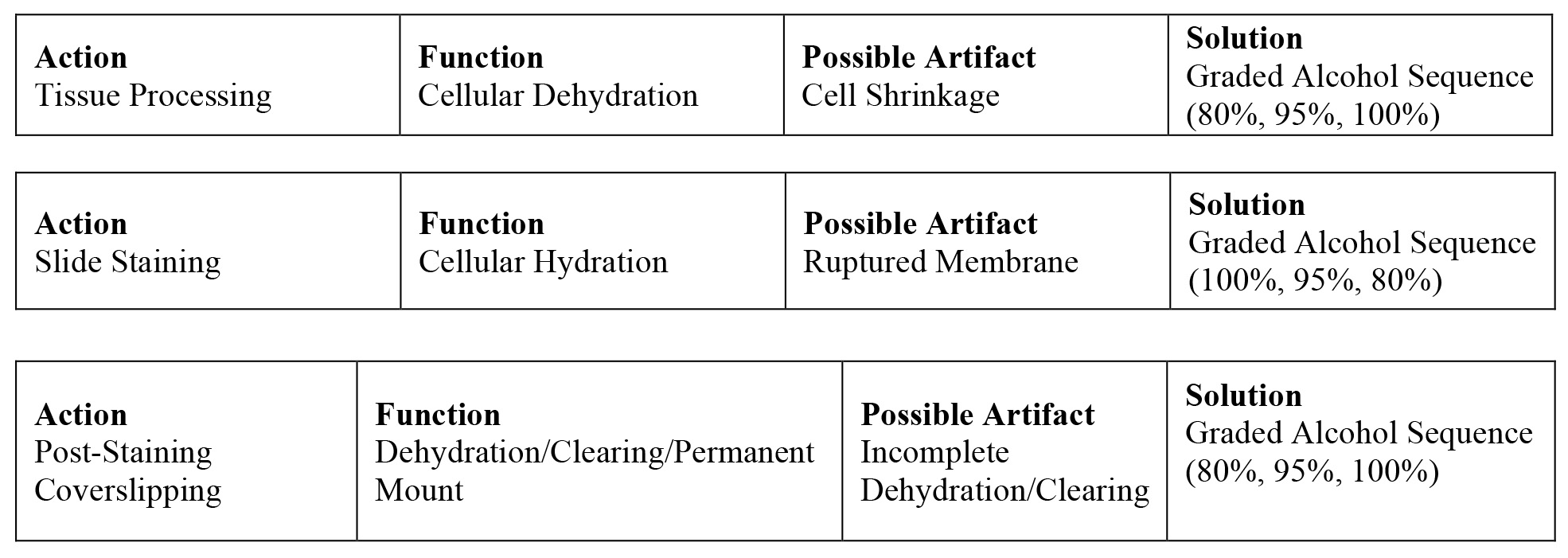
While this seems like a very elementary process of removing water in tissue processing, and replacing water in tissue staining, the success/failure and optimization of this goes back to the basic understanding of this semi-permeable cell membrane. Remember, the cell membrane regulates the flow of fluids in and out of the cell through the previously mentioned osmotic pressure. The membrane is a delicate structure that can expand or contract to accommodate the pressure within or outside the cell. If for example in tissue processing, the cells are subjected to a dehydration process that is too rapid for the cell membrane to manage, then a gross distortion of the membrane can occur. Alcohol is the most common dehydrant use in routine histology, however it produces an extreme hypertonic solution to the cell where it rapidly draws all of the bound water out of the cell. If the process is not controlled and managed, then it could result in rapid cell shrinkage and the morphology changing upon diagnostic examination to a wrinkled appearance. Tissue must never be placed immediately from the fixative (formalin) into 100% alcohol. Conversely, if rehydration of cells/tissue happens too rapidly in preparation for slide staining, then the intense pressure on the membrane from taking on water too quickly can cause the membrane to expand and rupture. The scenario in staining of going from deparaffinization in xylene, to clearing in 100% alcohol, then immediately to water, this forms an extreme hypotonic effect to the cell. This is particularly problematic in cytology where cells are suspended in a medium, as opposed to tissue with greater support. The top and middle tables below illustrate the necessity of using graded alcohol steps in both processing and staining. In the third table on the bottom, we see the post-staining coverslipping steps where a permanent mounting medium is used to fix a coverslip on the slide. This provides protection for the slide and facilitates long-term slide storage. The mounting medium used is typically a clear xylene or toluene-based medium that is miscible with the previous slide clearing step in xylene. After staining, if the dehydration steps are not optimal, water will carry over to the clearing steps in xylene, and the permanent mount (coverslipping) will have a cloudy appearance.
References
- https://www.genome.gov/genetics-glossary/Cell-Membrane.
- https://letstalkscience.ca/educational-resources/stem-in-context/osmosis-and-its-role-in-human-biology-and-health; “Osmosis and Its Role in Human Biology and Health”.
- one-school.net, “Cell, Cellular Components and Their Function”.