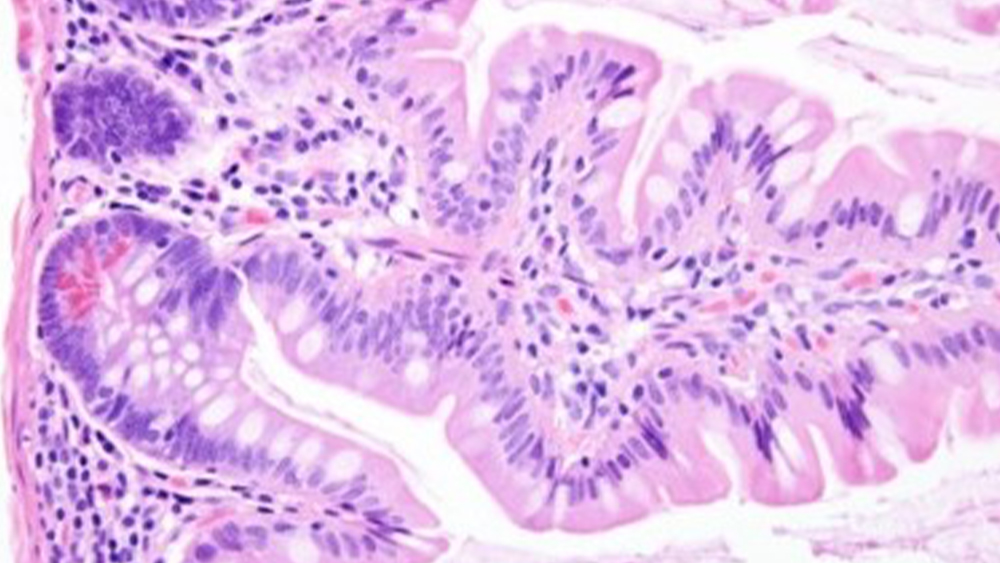
Eosin Y, unlike Hematoxylin which is a natural dye, is a synthetic dye and part of the xanthene family of dyes; a series of orange-pink to orange-reddish dyes. They include phloxine B, fluorescein, and Eosin B. Eosin Y is derived from fluorescein. Eosin is acidic and negatively charged. It will have an attraction to and bind with positively charged proteins in cytoplasm and connective tissues (muscle, cell membrane, etc.). Historically when Eosin first came into use it was as aqueous eosin. Eosin is very soluble in water. In staining, given that the tissue had been saturated with water from previous rinse steps, the eosin had less of a binding capacity with the tissue and also would immediately begin bleeding back out with subsequent rinse steps. It was very challenging to control and reproduce the level and intensity of stain from one stain run to the next. Also, it had a strong characteristic of continued leaching out of the issue after coverslipping. Further development of Eosin Y as a counterstain led to the development of alcoholic Eosin Y. Eosin Y’s solubility is 5-10 times greater in water than in alcohol, so an alcoholic content in the formulation increased the capacity for eosin to bind and maintain its attraction to positively charged tissues. Most Eosin Y formulations made in the U.S. today have a 60-80% ethanol component. This helps bring out its fluorescein derivative characteristic and give a greater differentiation of three shades of Eosin staining in tissue. Optimally, a good alcoholic Eosin Y should produce a stain intensity differentiation between red blood cells, collagen and cytoplasm, and muscle. Note the distinct red intensity of the red blood cells in the image on the far left; the softer hue of the cytoplasm providing a backdrop for nuclei in the center image; and the bold pink staining of smooth muscle cells with elongated nuclei in the far right.
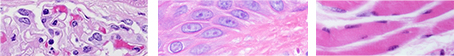
As previously mentioned, pH is a primary determining fact in the efficacy of Eosin Y as a counterstain. pH is critical to determining the charge of both the dye solution and the tissue molecules, and thus maintaining an attraction for each other. A low pH solution will increase the number of positively charged sites in the tissue. A high pH solution will decrease the number of charged sites and reduce the attraction of dye to tissue. While subordinate factors such as dye content of Eosin, staining time in Eosin solution, and excessive rinses require consideration, pH is the absolute number one factor that progressively changes with each staining run performed.
Alcoholic Eosin Y, as an acidophilic dye, has an optimal staining capacity at a pH of 4.3 to 5.0. With every introduction of new slides to stain in the same solution, more and more carryover of water is introduced into the solution. pH is stable to a point, but depending on the volume of Eosin Y solution in your staining dish, and the increasing volume of residual water from slides being stained, the pH will begin to rise above the optimal level of 5.0. The rising pH will progressively alter the binding capacity and overall performance of Eosin Y and should be monitored, particularly in high volume slide staining labs performing 500 slides or more. This is the primary benefit of the 95% alcohol rinse placed just prior to the Eosin staining step that was previously mentioned. The 95% alcohol step will remove most of the bound water in the tissue slides and increase their positive charge.
In the two images shown we see two like tissue sections of small intestine; the image on the left demonstrates alcoholic Eosin Y counterstaining at a pH level of 4.3. The image on the right demonstrates the same section stained at a pH level of 5.2. Note the considerable reduction in depth of pink staining; also, note the intensity of the red blood cells seen in the far-left portion of the image on the left, contrasted by the very weak staining on the right image. It is imperative that the pH is maintained in the optimal range.
References:
- Brown, S., “The Science & Application of H&E Staining”, Workshop in Technical Develop Series, Lab Management Consultants, 2011.
- Brown, S., “A Systems Approach to H&E Staining”, Workshop presentation, National Society for Histotechnology Symposium, 2013.
- Hansen, S., Images in H&E Staining, LBS, 2011.
- Guidelines for Hematoxylin & Eosin Staining, NSH, Bowie, MD, 2001.