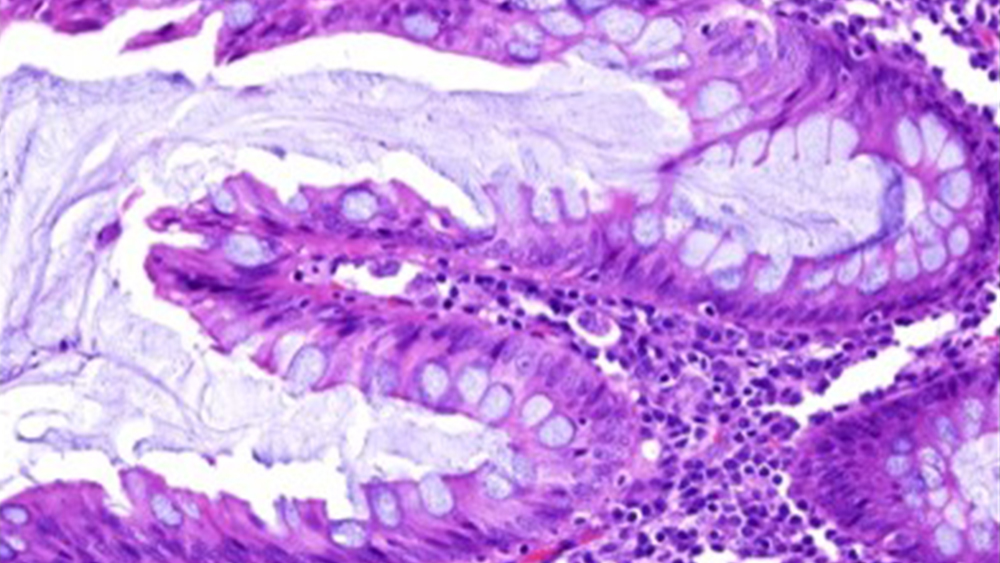
The Hematoxylin & Eosin (H&E) stain is the most common and universally known histology staining technique and is based on the study of nuclear v/s cytoplasmic staining in tissue. Specific dyes have an affinity for specific molecules in tissues. The concept of ‘acidophilic’ dyes (having a pH of ˂ 7), and ‘basophilic’ dyes (having a pH of ˃ 7), is the foundation of a dye’s specificity for tissue elements. The nucleus of a cell, which is the primary point of interest in pathology, has negatively charged molecules and will have a strong affinity for positively charged molecules in certain dyes.
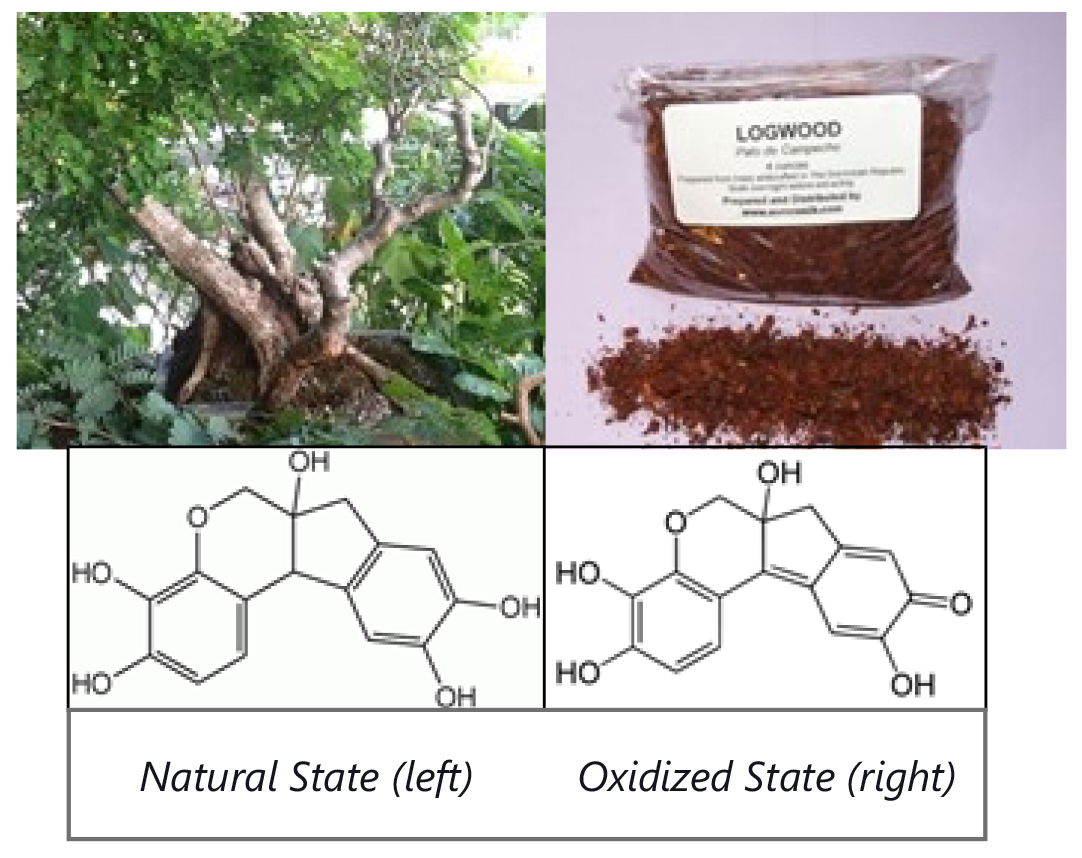
Hematoxylin is a natural dye extract which is formed in the Logwood tree found in Mexico and Central America. To date there has been no other substance or source found to provide the quality and staining morphology better than hematoxylin. Hematoxylin in itself has little to no staining capacity. When oxidized it forms a substance called ‘hematein’. The chemical compound images illustrate this process. Hematein, then bound to aluminum, forms an overall positive charge. The chromatin material found in the nucleus of cells is strongly negative in its charge. So now you can see the strong affinity of a positively charged hematein/aluminum compound to a negatively charged nuclear chromatin. This gives us the richness and depth of the hematoxylin dye.
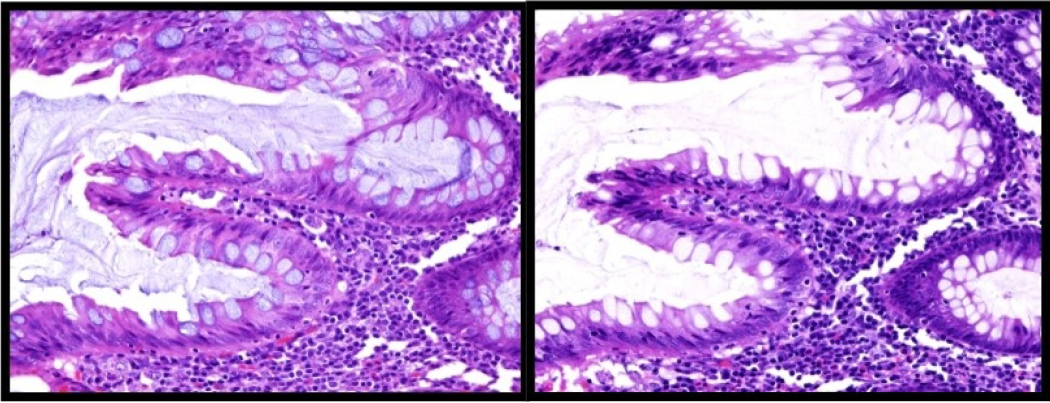
Hematoxylin gives a natural staining response of reddish-purple and after staining must be treated with an alkaline rinse to bring out the deep bluish-purple color. This post staining step is called ‘bluing’. The pH of this rinse is typically 7.6 – 9.0. With the progressive hematoxylins oftentimes tap water can be used for this step because it is on the average about 7.5 pH, however, tap water is very inconsistent from region to region based on the local water treatment methods. It is not recommended for regressive hematoxylins.
As mentioned in the beginning of this article, the hematoxylin stain is a basophilic dye with an affinity for negatively charged molecules. It has an overall positive charge, but when it becomes diluted from repeated staining steps (either by water or depleted dye content), the stain will begin to rise in pH and thus lose its affinity for nuclear chromatin. Slide staining quality should always be monitored when doing multiple staining runs through the same hematoxylin solution. Staining methods should be developed and validated based on, among other things, the staining capacity of the hematoxylin.
In this article we have given you some basic but essential information on hematoxylin that will assist you producing distinct, crisp nuclear detail in your staining. In part 2 we will discuss Eosins and the balance of coloration between nuclear and cytoplasmic staining.
References:
Brown, S., “The Science & Application of H&E Staining”, Workshop in Technical Develop Series, Lab Management Consultants, 2011.
Brown, S., “A Systems Approach to H&E Staining”, Workshop presentation, National Society for Histotechnology Symposium, 2013.
Hansen, S., Images in H&E Staining, LBS, 2011.
https://www.theyucatantimes.com/2016/08/palo-de-campeche-logwood-natural-dye-that-changed-the-textile-industry/